 P(NH3).NH3(g) + H+ + Cl- → NH4+ + Cl-Īll added entities should sum to zero if the cyclical process is reversible, no? 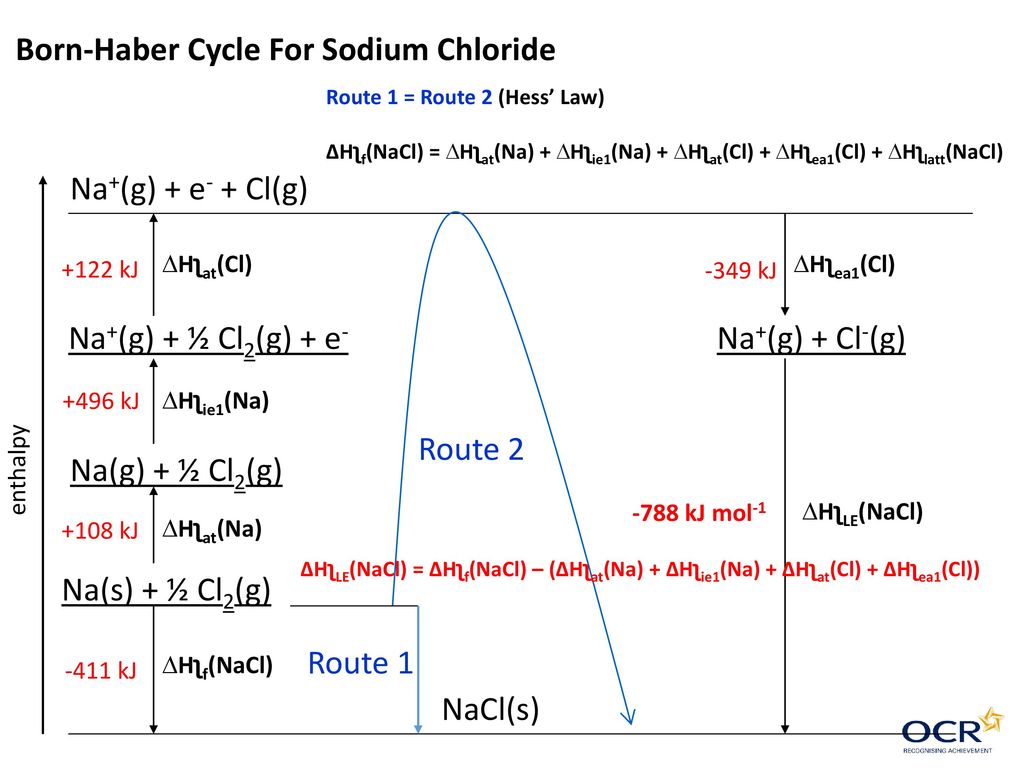
Lattice energy/mole NH4CL U = 640kj/mol, ionization energy/mole hydrogen atoms IH = 1305kj/mol, electron affinity/mole ECL = 387kj/mol, heat of formation for NH3 = -45.6kj/mol, heat of formation for NH4Cl = -314.2 The Born Haber cycle for the process was given as follows: 
The problem statement, all variables and given/known d 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
